
T-CELL ACTIVATION
Atract™ T-Cell Activation Kit
T-cell activation reagents, based on non-magnetic beads, bound with antibodies.
KEY FEATURES:
- Magnet-free activation
- No residuals - no downstream cleaning required
- Non-phagocytisable particles
- CD3/CD28 and CD2/CD3/CD28 version
- Highly efficient CAR transduction
- Great for T-cell assays with no debeading needed
- High and consistent T-cell activation frequency
- Scalable across flasks, bags, plates and G-Rex® platforms
Integrate the Atract™ T-Cell Activation Kit into your workflow now!
REQUEST A QUOTE
Atract™ T-Cell Activation Kit V1 (CD3/CD28)
Functionalised with anti-CD3 and anti-CD28 antibodies for effective T-cell activation and expansion.
Atract™ T-Cell Activation Kit V2 (CD2/CD3/CD28)
Functionalised with anti-CD3, anti-CD28, and anti-CD2 antibodies, designed to establish a superior proliferative capacity of stimulated cells.
| ATRACT™ VERSION | ANTIBODIES | FORMAT | ACTIVATION CAPACITY | CATALOGUE NUMBER |
|---|---|---|---|---|
| Atract™ Kit V1 | Anti-CD3/CD28 | 4x 10 mg | Up to 12*10^6 PBMCs | 3830077790200 |
| 4x 50 mg | Up to 60*10^6 PBMCs | 3830077790194 | ||
| Atract™ Kit V2 | Anti-CD2/CD3/CD28 | 4x 10 mg | Up to 12*10^6 PBMCs | 3830077790156 |
| 4x 50 mg | Up to 60*10^6 PBMCs | 3830077790088 |
MAIN BENEFITS
REDUCE COST WITH EFFICIENT ACTIVATION
Atract™ triggers robust T-cell activation and high proliferation. This lowers the cost of the cell activation step, which in turn means lower consumable expenses, facility usage and overall process costs.
CELL THERAPY ORIENTED PRODUCT
Cells stimulated by Atract™ Kit exhibit a favourable phenotypic profile, low exhaustion marker expression and highly efficient CAR transduction. Integration of Atract™ into workflows can thus upgrade the production process and help foster a more potent final therapeutic product.
SAVE ON LABOUR TIME
Atract™’s simple, scalable protocol integrates into existing systems, including flasks, plates, bags, and the G-Rex® platform. Customers can scale output without redesigning their process, reducing personnel time, training needs, and tech transfer complexity.
ATRACT™ = NO RESIDUALS
REDUCE DOWNSTREAM STEPS
Atract™ is designed to deliver clean, efficient T-cell activation without introducing unwanted impurities into your workflow. Unlike conventional activation reagents, Atract™ reagent particles cannot be phagocytised, meaning they do not get taken up by cells and are therefore easy to remove from the culture after stimulation.
This residual-free approach helps eliminate the need for additional downstream cleaning steps, keeping handling to a minimum and supporting simpler, more cost-effective processes.
Atract™ Kit is fully compatible with standard and scalable cell culture formats, including conventional flasks, bags, plates, and G-Rex® platforms, making it suitable for both research and manufacturing environments.
Supplied in a biocompatible, azide-free buffer, Atract™ can be used directly for cell stimulation without additional preparation—enabling faster, cleaner, and more streamlined workflows.
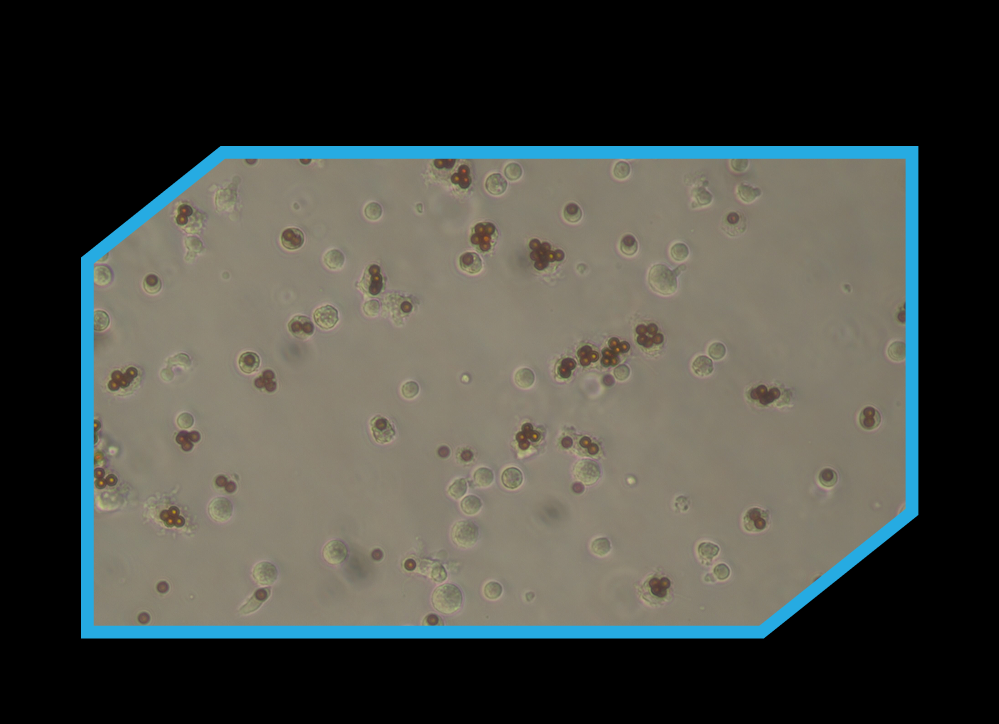
Competitor
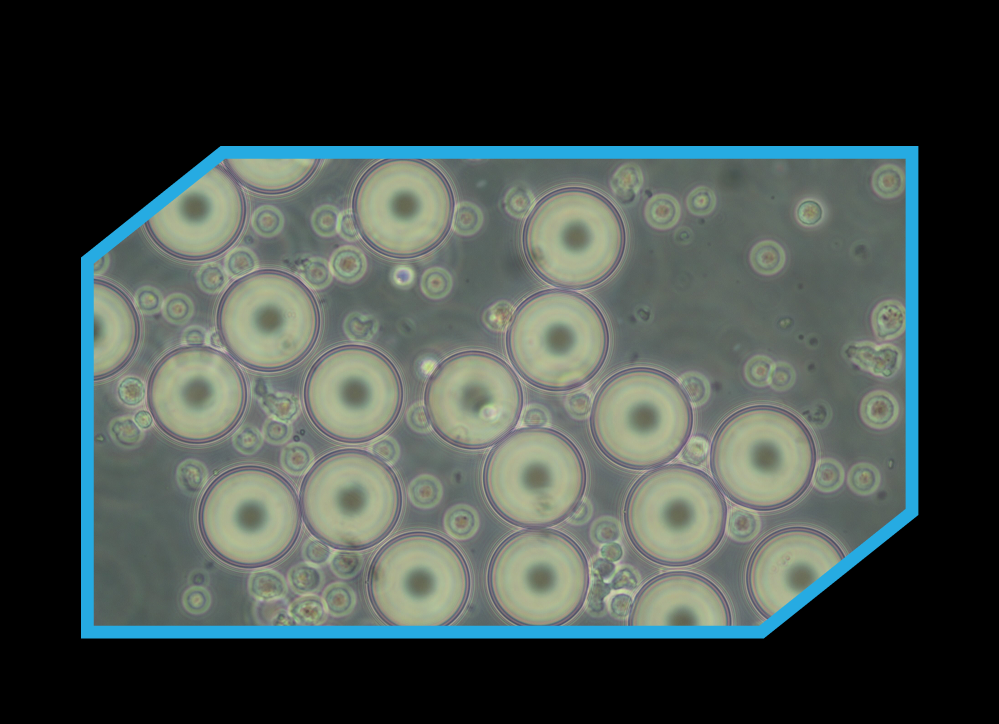
Atract™ Kit

ATRACT™ KIT IN PRACTICE
Straightforward, cost-efficient scaling: the required amount of Atract™ Kit is easily calculated based on culture surface area, enabling predictable scale-up without additional optimization or material waste.
Cell-friendly activation with less handling: Atract™ Kit particles sediment to form a stable activation surface, efficiently stimulating cells while preserving viability and proliferation, reducing hands-on time.
Clean removal, simplified downstream steps: the reagent can remain in culture or be easily removed using standard 30 μm filtration, delivering pure cells without residual particles that could affect product quality or regulatory compliance.
Consistent, high-quality cell output: T cells show high viability, sustained activation marker expression, low exhaustion markers, and a favorable metabolic profile after activation and expansion.
RESOURCES
FAQ
Which samples are appropriate for activation with Atract™?
Is Atract™ reagent removal necessary?
Which 20-35 μm strainers are suitable for filtering the suspension?
Can we use different protocols and adjust the official protocol?
Are there any requirements for cell culture containers?
Why does the protocol suggest adjustments for 96-, 48-, 24-, and 12-well plates?
ORDER